 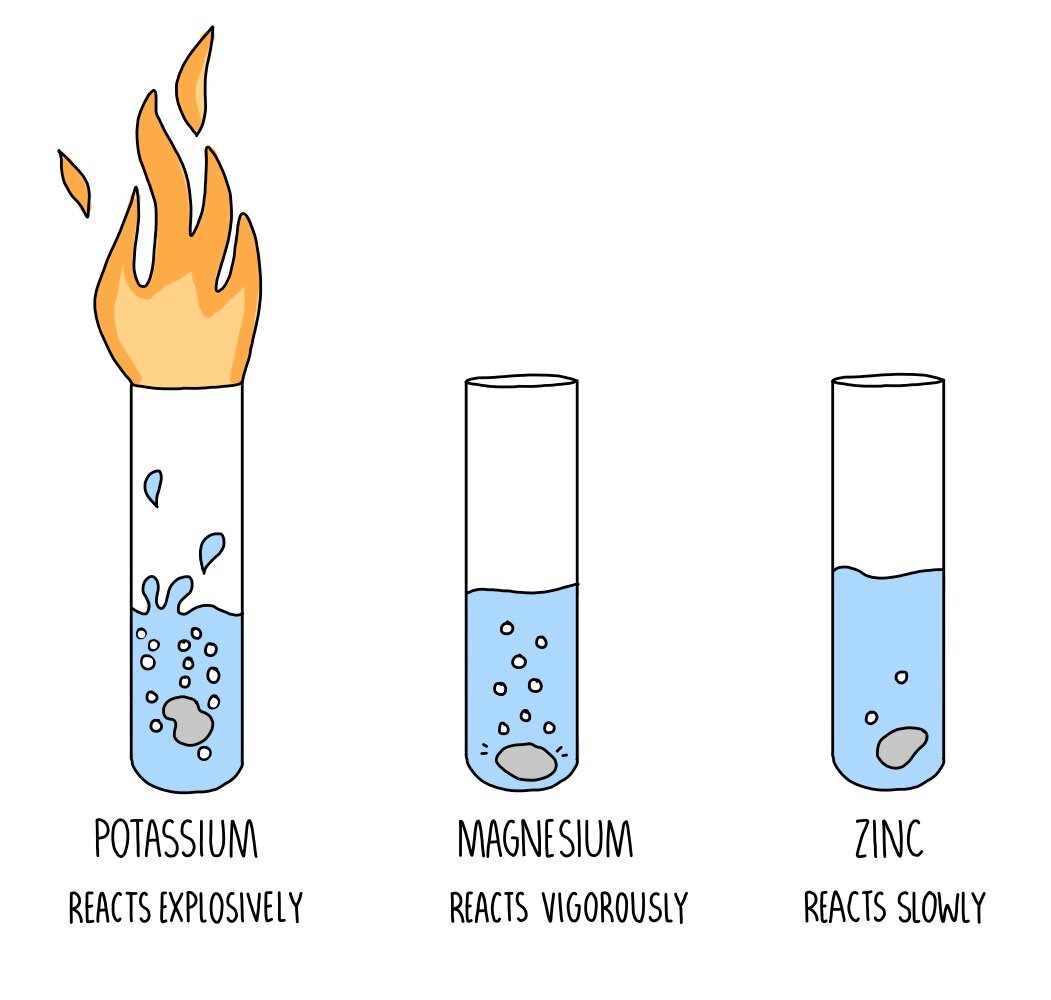
When a metal reacts with cold water or steam, hydrogen is always produced. Oxides are formed when metals react with steam. Hydroxides are formed when metals react with cold water. Despite the fact that calcium, strontium, and barium hydroxides are only marginally water-soluble, they produce enough hydroxide ions to make the surrounding environment basic. Metal oxide is formed when a metal, such as magnesium reacts with steam as a result of metal hydroxides breaking when heated. Metal hydroxide is formed when a metal reacts with cold water. Magnesium has a minor reaction with water, but when it is combined with steam or water vapour, it produces white magnesium oxide and hydrogen gas. Beryllium also has a tough outer oxide layer that restricts its reactivity at low temperatures. Beryllium (Be) is the only alkaline earth metal that, even when heated to red heat, does not react with water or steam. Similar to Group 1 metals, alkaline earth metals (Be, Mg, Ca, Sr, Ba, and Ra) are the second most reactive metals in the periodic table, increasing reactivity as the period increases. As the periodic table progresses, the alkali metals, also known as Group 1 metals, become more reactive. The Group 1 metal (M) is oxidised to metal ions, and water is reduced to hydrogen gas ( H₂ ) and the hydroxide ion (OH). The alkali metals (Li, Na, K, Rb, Cs, and Fr) are the most reactive metals in the periodic table they all react quickly, if not explosively, with cold water, displacing hydrogen. 
Less reactive metals are displaced from their complexes as more reactive metals react with water. The reactivity series ranks metals according to how easily they react. Acid-resistant gloves and a face shield are required, and the procedure should be performed in fume hoods. Some water-reactive chemicals, such as organometallics and sulfuric acid, are also pyrophoric and should be kept dry. Alkali metals, which range from sodium to caesium, and alkaline earth metals, which range from magnesium to barium, are two prominent examples. Water-reactive compounds strongly reduce chemicals that undergo a chemical reaction with water on their own. The higher the electronegativity, the higher you move up and to the right on the periodic table. The greater the electronegativity, the more active the electron exchange. As we move through the group, our response rate decreases. Nonmetals – Our reactivity increases as we move from left to right. Metals a period reactivity decreases as you move from left to right over time.Īs you move further to the left and down the periodic table, the higher the reactivity, the easier it is for electrons to be given or taken away. Because chemical processes rely on electron transfer/interaction, this is primarily defined by how easily electrons can be removed (ionisation energy) and how urgently they want to take the electrons of other atoms (electronegativity). 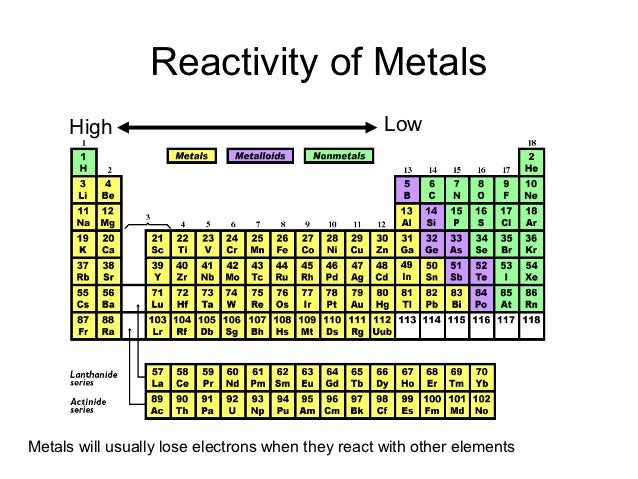
Reactivity refers to the likelihood or intensity with which an atom reacts with other substances. Trends in chemical reactivity with water Chemical reactivity 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
