 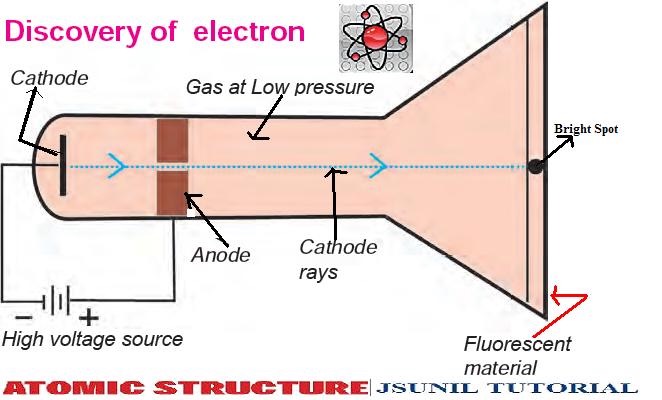
Starke observed the differences in the mechanical reaction for strong and weak electrical currents, changes in vacuum pressure, metallic and non-metallic surface materials, and different angles of incidence of the beta rays on the surface, and concluded that thermal effects were greater than the mechanical impulse imparted by the electrons. Starke "Notiz über die mechanische Wirkung der Kathodenstrahlen" Annalen der Physik 3, (1900) pages 101-107. Thomson "Conduction of Electricity Through Gases" Cambridge University Press 1903 p.501ģH. The voltage sources cause one disc to become positively charged. The metals get connected to a voltage source. The tube is a glass chamber with two metal discs. It's creatively called a cathode ray tube. Cathode rays get produced by a unique apparatus. Thomson performed three experiments with cathode ray tubes. The tube is only about 12" long and the paddle wheel a few centimeters in diameter, so video projection should be used when presenting to a large audience.ġSir William Crookes "On Radiant Matter" Popular Science Monthly, London 1880 p.158ĢJ. In the 1890's JJ Thomson investigated cathode rays. Great care must be taken to make sure the tube is level and the frictional forces on the axle of the paddle wheel can be overcome. In the presence of electric and magnetic fields, the cathode rays are. Use the same 6V battery and induction coil used for the Maltese Cross and Cathode Ray Deflection Crookes tubes. Thomson made a remarkable improvements in the scope of study of gases in discharge tubes. Please use gloves and safety glasses when handling this fragile vacuum tube. Starke, showed that momentum transfer of the electrons was not enough to account for the observed motion of the paddle wheel, and concluded that the kinetic energy of the electrons only indirectly leads to movement of the paddle wheel via radiometric effect. Thomson, citing the experimental work of H. In 1880 Sir William Crookes argued that the mechanical impulse of the electrons hitting the vanes caused the observed motion-similar to how flowing water turns the arms of a turbine. Flourescent material on the vanes glow green when subjected to the energetic electrons. This ushered in a model of atomic structure referred to as the plum pudding model. Dalton thought that atoms were indivisible particles, and Thomson's discovery of the electron proved the existence of subatomic particles. Your job is to measure the deflection of the beam under different conditions and then determine the ratio of the charge of the particles in the beam to the mass of the particles in the beam. Thomson's experiments with cathode ray tubes helped him to discover the electron (which Dalton did not know about). When 40kV is applied across the tube, the lower vanes directly between the cathode and anode pivot away from the cathode, causing the wheel to move. In this lab we will be looking at a model of the Thomson experiment using a Cathode Ray Tube. A beam of cathode rays (electrons) impinging on a paddle wheel cause it to spin and travel down the vacuum tube.Ī paddle wheel is suspended by its axle inside a Crookes tube so that when the paddle vanes spin the entire wheel is free to travel the length of the tube. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
